Forschungsbereich - Photoelektrochemie
In diesem Forschungsbereich beschäftigen wir uns mit licht-induzierten Prozessen in elektrochemischen Systemen für unterschiedliche Fragestellungen aus dem Bereichen der Elektrodeposition oder auch der Energiespeicherung und -wandlung. Unter anderem betrachten wir hierbei die Kombination von Photovoltaik und Elektrolysezellen als auch dem integrierten photoelektrokatalytischen System, in dem direkt licht-induzierte elektrokatalystische Reaktionen ablaufen, z.B. Wasserspaltung zur Erzeugung von Wasserstoff als Energieträger.
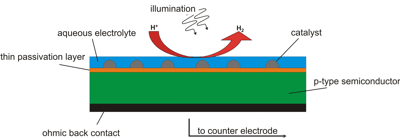
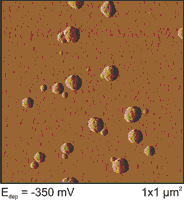
The chemical modification of semiconductor electrodes plays a key role in designing an efficient energy converter. For the light-induced hydrogen evolution reaction (HER) thin metal overlayers are important catalysts. Hence, a controlled deposition of the metal onto the semiconductor electrode is important to ensure optimal catalyzing of the HER. Photoelectrodeposition is investigated by cyclovoltammetry (CV) and atomic force microscopy (AFM). The variation of deposition parameters (e.g. time and/or overpotential) leads to different cluster densities and size distributions which affects the activity of the photocathode.
&nbs
