Measurement of neuronal activity with combined multifunctional micro NMR probes and micro electrodes
Conventional fMRI experiments are based on the BOLD effect, a local neuronal activity induced change in blood oxygenation, which allows for the detection of neuronal stimulation through the corresponding local susceptibility changes. This change in local susceptibility is usually measured with T2* sensitive EPI sequences with a temporal resolution in the range of seconds and spatial resolutions of several microliters. The proposed research projects aims to overcome these limitations in spatial and temporal resolution with the goal of assessing finer details of the basic neurovascular coupling cascade.
The main goal of this project is the detection of neuronal activity-related changes of proton magnetization at a significantly improved temporal resolution and sensitivity within small volumes of a few nanoliters accompanied by simultaneous recording of electrical activity. In addition, we will assess local changes of neurotransmitters such as Glutamate, Glutamine and GABA using proton spectroscopy with intrinsic volume selection, as well as energy metabolism via 31P spectroscopy.
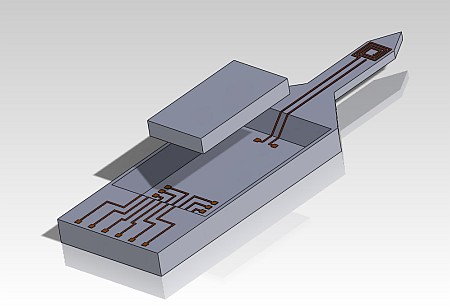
To achieve these goals, a microsystem consisting of a miniaturized MR coil, microelectrodes for neuronal recording and a custom designed ASIC mounted on a common needle-shaped substrate will be developed. The ASIC will perform all signal conditioning tasks for both the MR and the electrical signals in order to provide robust signals at its output which can be digitized using commercially available ADCs. In addition to the ASICs which will only perform the aforementioned signal conditioning tasks, TX/RX ASICs which can additionally provide the RF current necessary to excite the spin ensemble will also be designed. This local excitation will mitigate all problems associated with RF current induced tissue heating.
Overall, the envisioned system will be able to capture localized activity within single layers and preferably within regions of different cellular components (dendrites, bodies, etc.). In combination with electrical recording, direct study of neurovascular coupling can be performed at high temporal and spatial resolution to estimate the local dependence of hemodynamic response functions. In addition, the envisioned miniaturized multimodal recording system will allow assessing neurovascular coupling on an exceptionally fine time scale enabling to resolve correlations between electrical signals and proton magnetization changes far below the commonly assumed time lag of several seconds (fast coupling).
Project member
M.Sc. J. Handwerker
