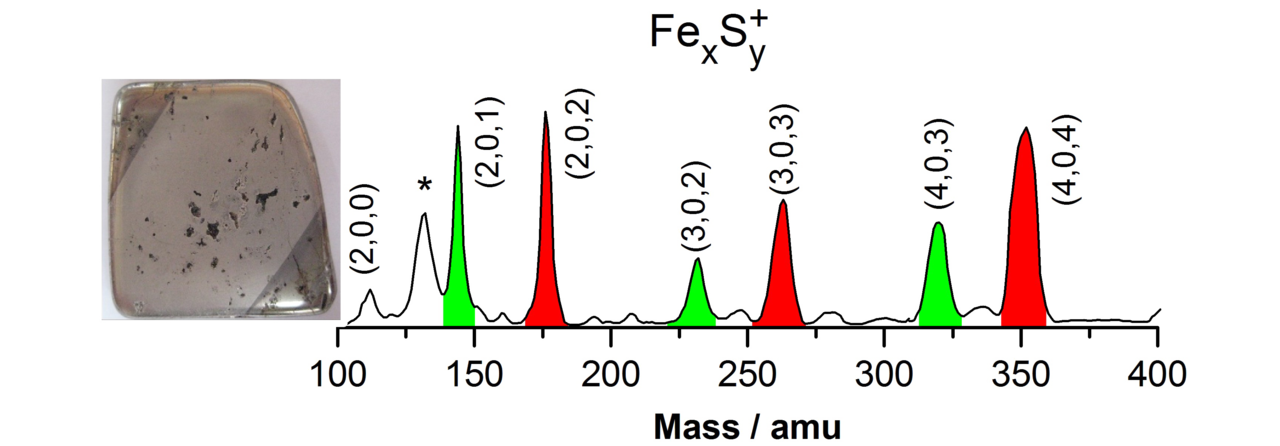
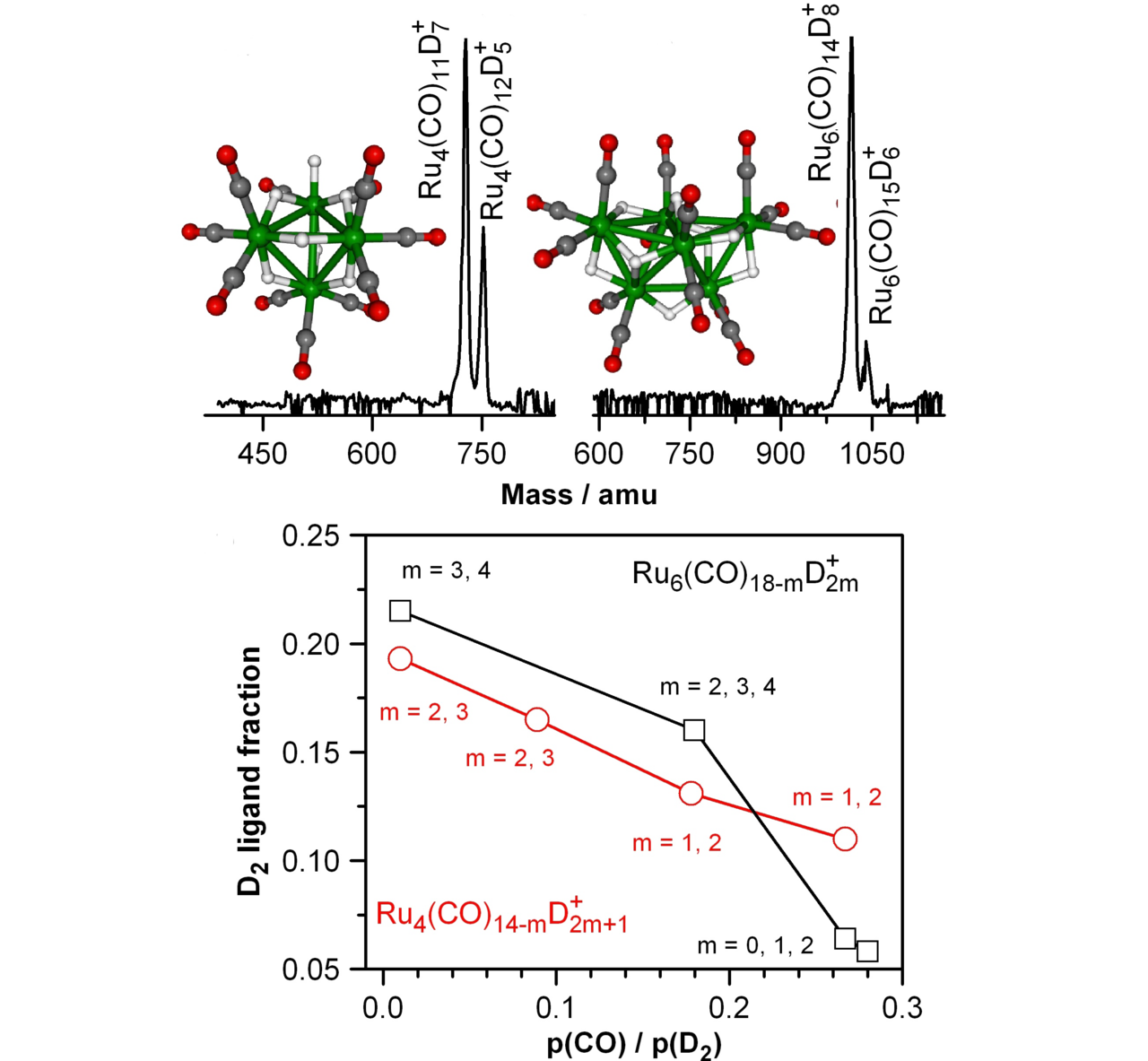
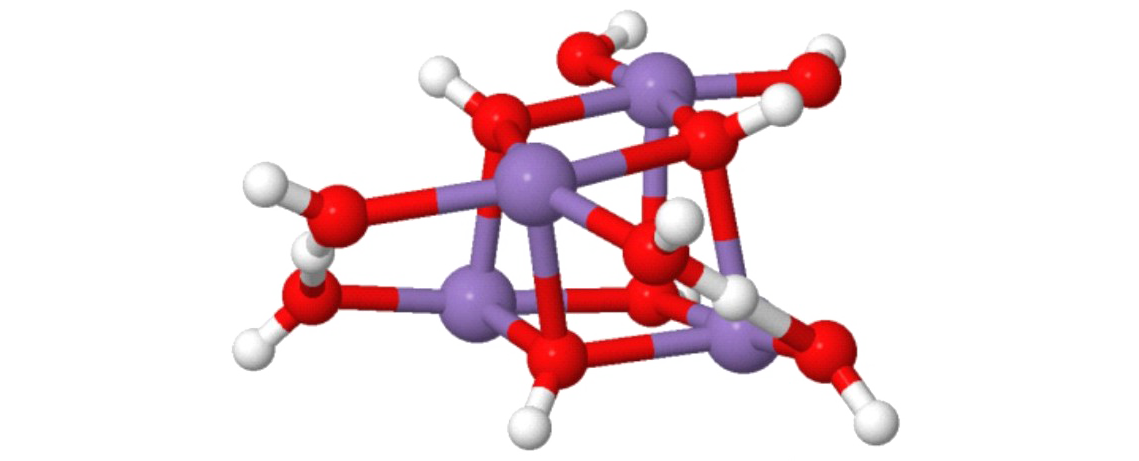
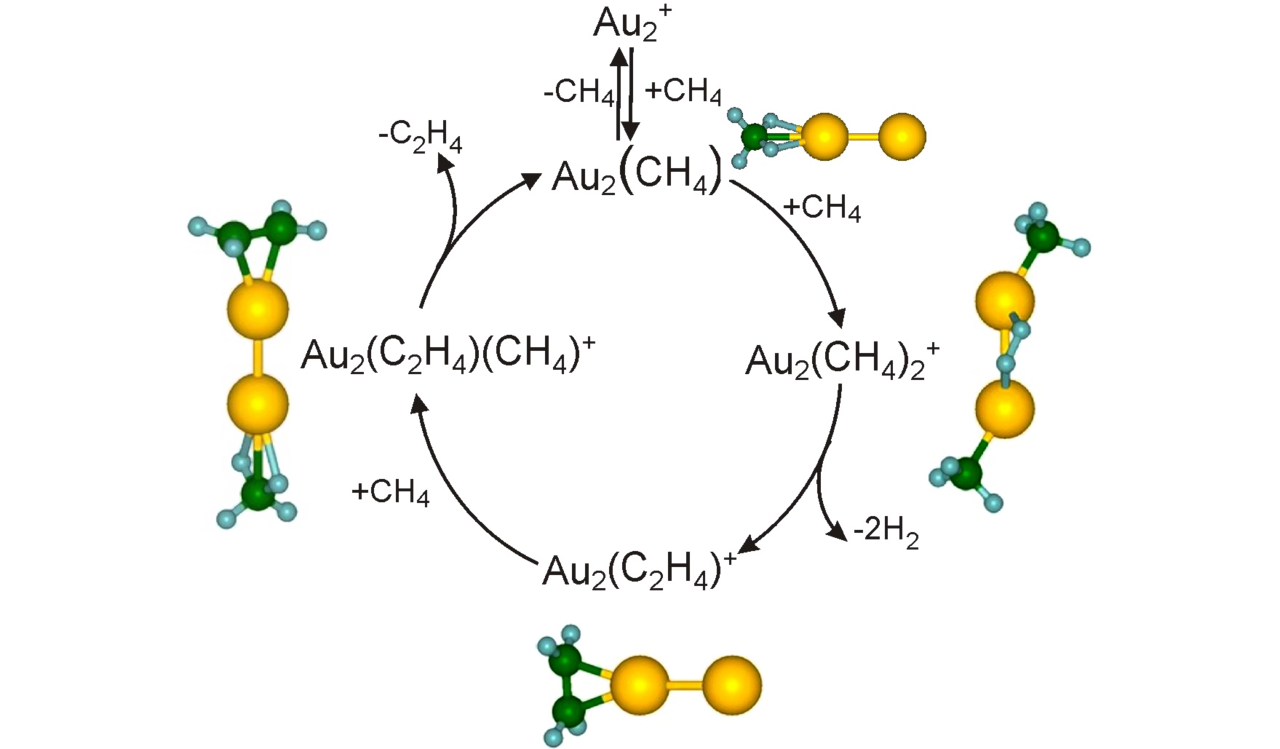
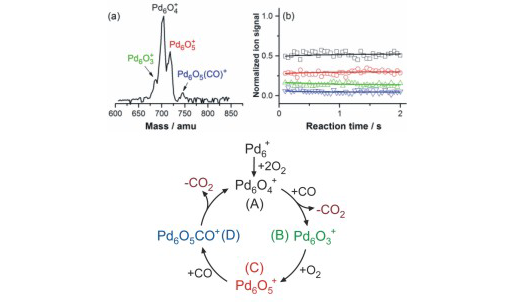
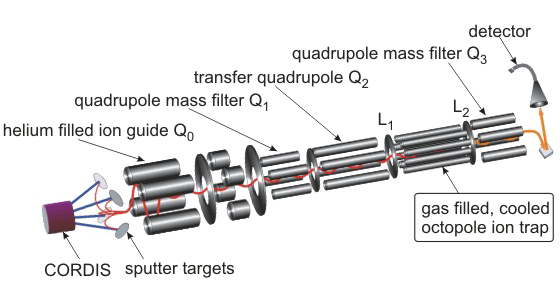
Small metal, metal-oxide, and metal-sulfur particles often represent the catalytically active centers (i.e., the location at which the actual chemical transformations take place) of heterogeneous and homogeneous catalysts as well as biocatalysts. In oder to desgin new tailor-made effective, selective, and energy efficient catalytic materials for specific reactions it is mandatory to understand the elementary processes of the catalytic reactions on a strictly molecular level. Towards this goal we employ small free clusters as model systems to mimic the catalytically active centers. Whith this approach we aim to gain fundemental insight into the kinetics and energetics of catalytic reactions as well as to elucidate bond activation/formation and charge transfer processes on a molecular level. In particular we focus on the influence of cluster specific properties (cluster size, charge state, composition, ligands) on the reactivity, catalytic activity, and selectivity. Furthermore, we are interested in coadsorption phenomena which often represent the first and rate-determining reaction steps in catalytic reactions and might even lead to catalyst poisoning.
More details can be found in:
- Phys. Chem. Chem. Phys. 14, 9255 (2012)
- Bunsenmagazin 15, 283 (2013)
- Chemical reactivity and catalytic properties of size-selected gas phase metal clusters, in ‘The chemical physics of solid surfaces (Vol. 12): Atomic clusters: From gas phase to deposited’, edited by D. P. Woodruff, Elsevier (2007).
- Gas Phase Model Systems for Catalysis - GPMC 2018