B05: Circular RNAs modulate neuronal vulnerability, neuroinflammation and regeneration and are affected by TBI comorbidities
PI: F. Roselli
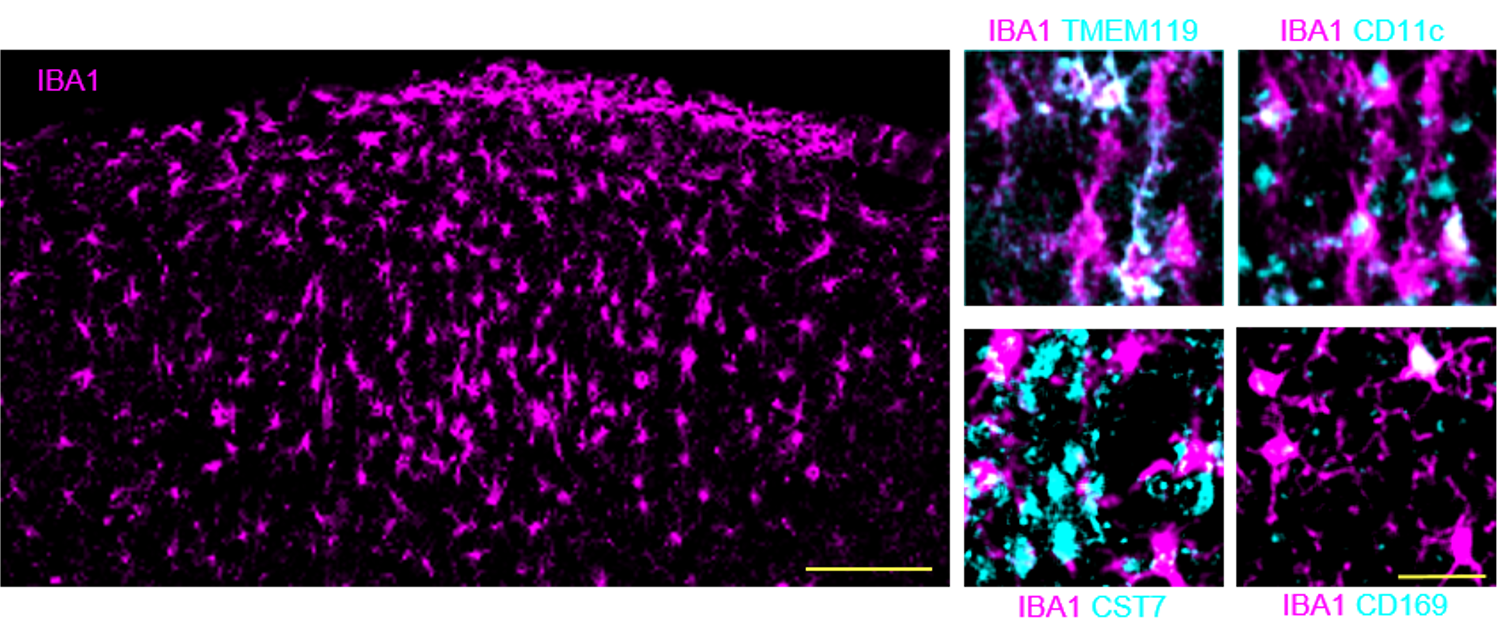
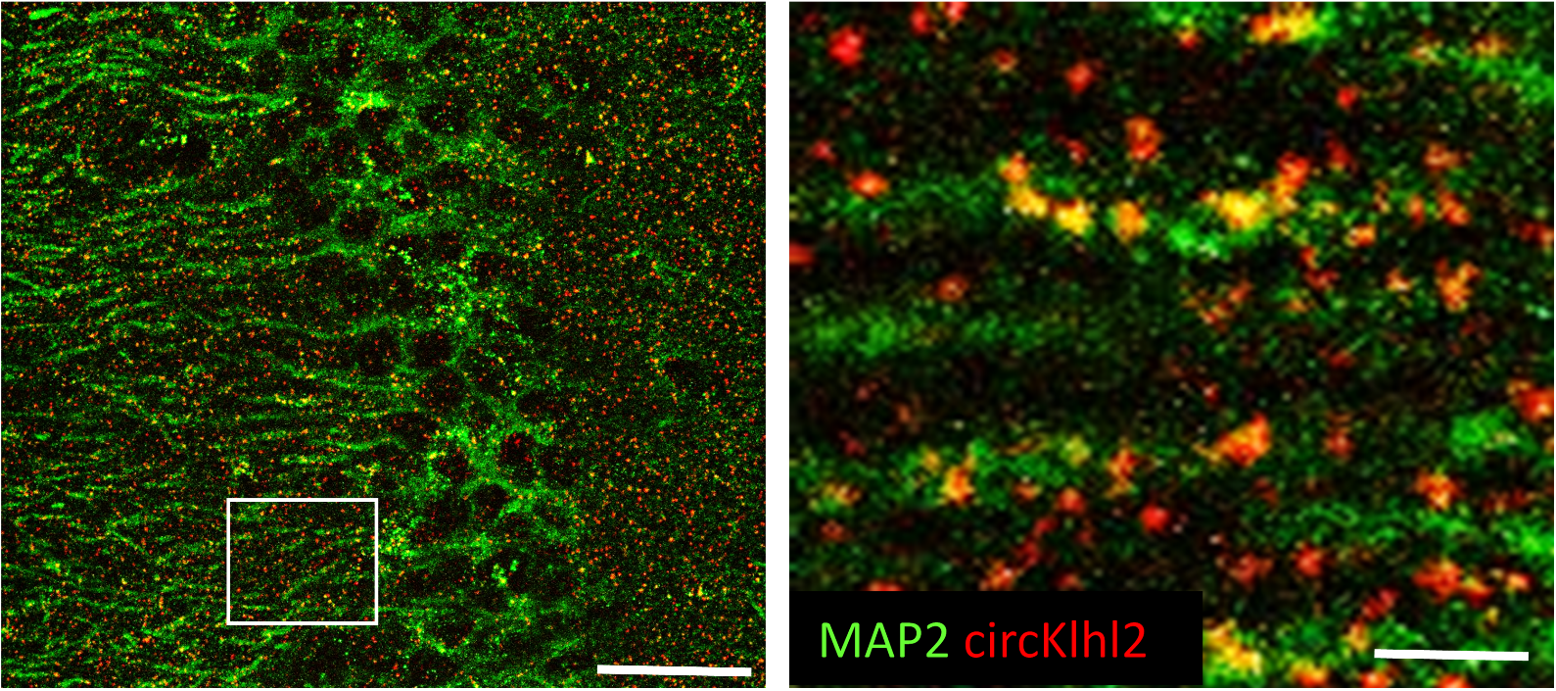
In the first funding period we revealed the neuroprotective and the neuroimmunomodulatory effects of ethanol intoxication (EI) concomitant to Traumatic Brain Injury (TBI). In the second funding period, we performed mechanistic investigations of EI, showing that EI-induced upregulation of IL-13/STAT6 increases neuronal activity and reduces microglial responses. Decreased activity-related nuclear Ca2+ signals caused the upregulation of microglial reactivity by suppressing neuronal Osteoprotegerin (OPG), leading to extensive synaptic loss. Thus, integrity in synaptic networks and sustained physiological neuronal firing are previously unappreciated determinants of neuronal vulnerability and neuroinflammation. Now we address the TBI role of circular RNAs, a new class of molecular regulators of synaptic networks. CircRNAs are non-coding RNAs endowed with wide-ranging regulatory effects through microRNAs and RNA-protein complexes. We show that several circRNA related to synaptic proteins are upregulated in murine and human TBI. We show that overexpression of circKhlh2, among the most abundant upregulated circRNAs, accelerates the recovery of spatial memory in mice. We plan to determine the effect of circKhlh2 on neuronal vulnerability, synaptic integrity and neuroinflammation in cortex and hippocampus upon TBI. Next, we will investigate the molecular mechanisms connecting circKhlh2 to microRNA abundance and to key neuroprotective and neuroplasticity mechanisms. Ultimately, we will explore how circKhlh2 expression upon TBI is modified by known comorbidies of trauma such as gender, age and EI. Finally, we set out to explore if CSF levels of synaptic proteins and of circKlhl2 are prognostic biomarkers in human TBI patients.
Principle investigator
- Prof. Dr. med. Dr. rer. nat. Francesco Roselli
Clinic of Neurology
Ulm University Medical Centre
Helmholtzstr. 8/1
89081 Ulm
Tel.: +49 731 500 63147
Francesco.Roselli(at)uni-ulm.de