Dynamic Behavior of Methanation Reactors
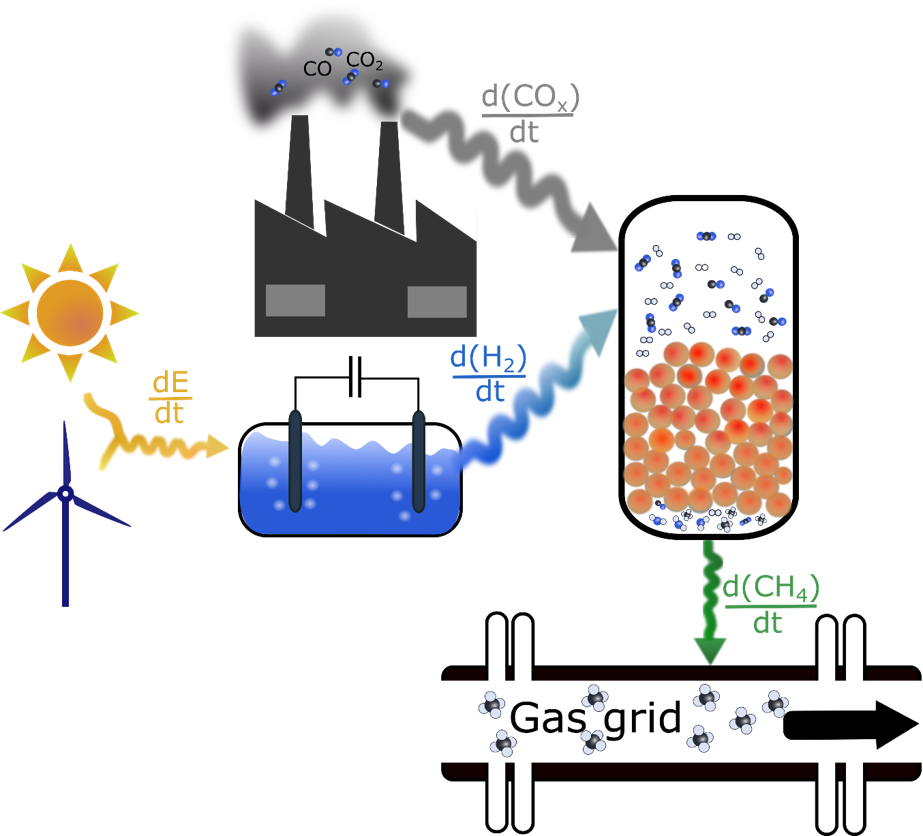
The operation of chemical reactors under unsteady-state conditions is getting more and more important, due to the implementation of renewable ressources into the chemical value chain. This induces dynamics at different length scales inside of the reactor, such as at the active surface, inside the catalyst pellet and at reactor scale, which are coupled with each other. The interplay of the respective kinetic processes determine the dynamic reactor response upon transient changes of inlet and operations conditions, which is challenging to be predicted for realistic reactions. This project aims at decoupling the dynamics of the involved kinetic processes by experimental methods for methanation of CO/CO2-mixtures as an example.
Contact: M.Sc. Alexander Zirn, M.Sc. Franz Braun
Collaborators: Prof. Dr.-Ing. Thomas Turek
PICASO - Intensified Ammonia Synthesis
Ammonia is one of the most produced chemicals in the world. It is not only an important bulk chemical but also extremely interesting as a vector for transporting green hydrogen. While conventional processes usually depend on natural gas as a hydrogen source, renewable Power-to-Ammonia processes use hydrogen from electrolysis. Due to its high purity, it enables the use of advanced catalysts which are highly active under much milder reaction conditions. Those are favourable for a reaction otherwise strongly limited by thermodynamic equilibrium, and with additional in situ product removal of the produced ammonia, the limitations might even be overcome. In the PICASO project, we aim at understanding and optimizing this intensified process and its dynamics resulting from both the use of renewable hydrogen and a solid sorbent for ammonia removal by experimental and theoretical investigations.
Contact: M.Sc. Theresa Kunz, M.Sc. Thomas Cholewa
Collaborators: Thermochemical Processes Department, Fraunhofer ISE
Funding: German Federal Environmental Foundation (DBU); Federal Ministry of Education and Research (BMBF) via Project Management Jülich (PtJ).
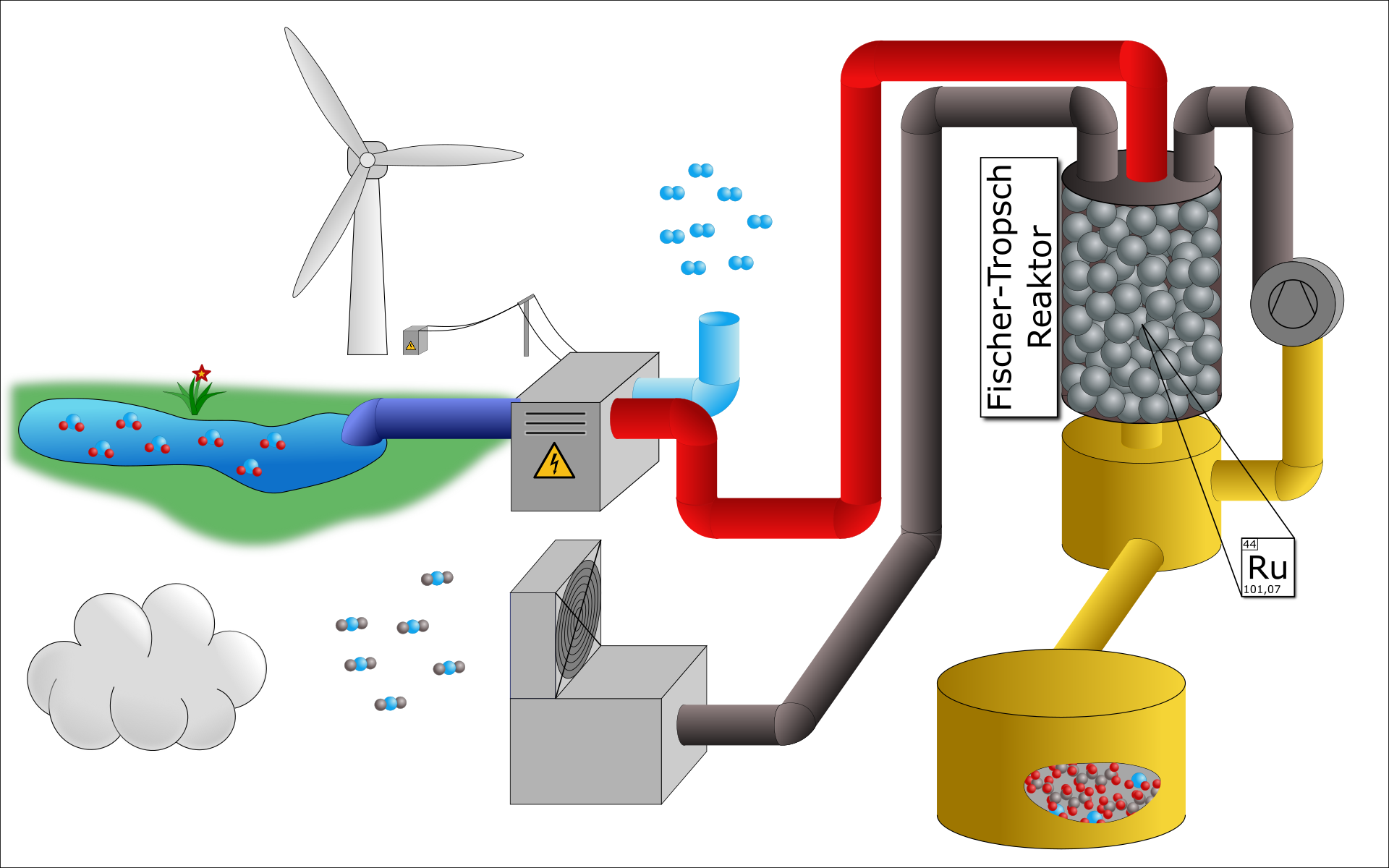
Bi-Functional Catalysts for Fischer-Tropsch-Synthesis
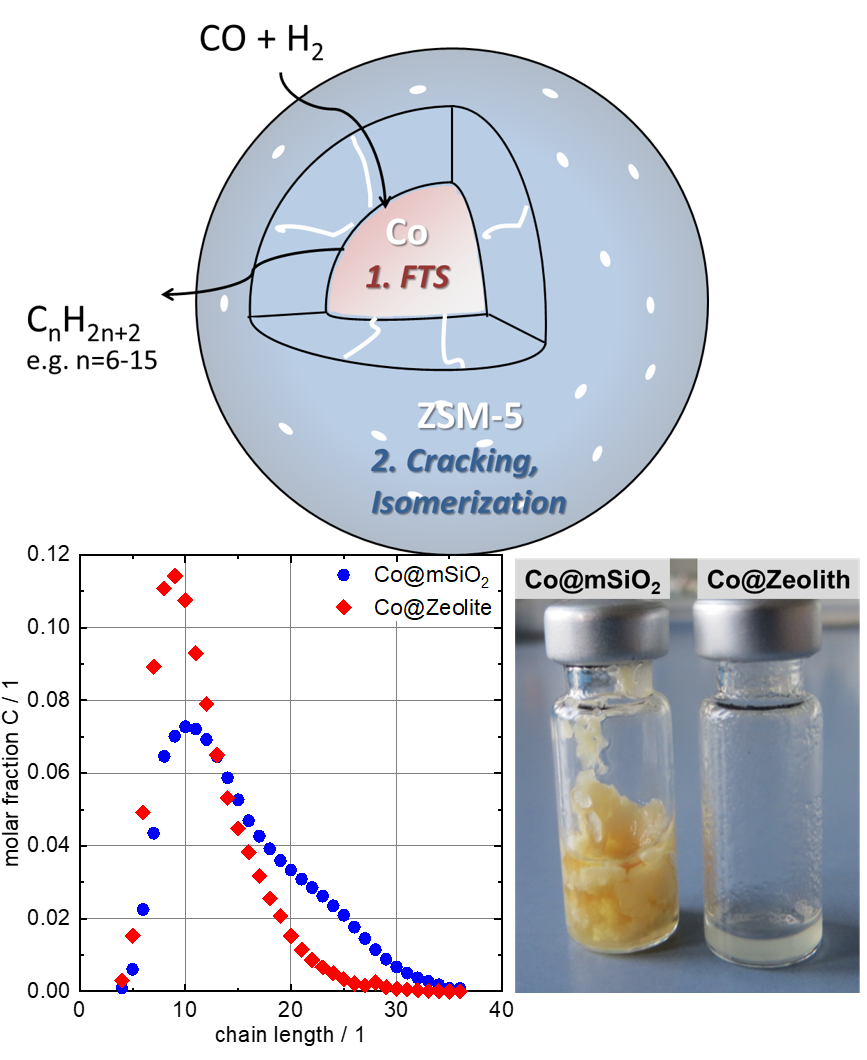
Fischer-Tropsch (FT) synthesis is an interesting process to convert renewable ressources into fuels and chemicals, as it allows to convert CO derived from biomass and waste gasification with H2 from water electrolysis towards hydrocarbons. Even though the process is implemented on industrial scale already, it suffers from a broad product distribution, which requires elaborate processing to meet the desired product specifications. Bi-funtional catalysts, combining a FT-active with a hydroprocessing component, offer the possibility to tailor the product distribution. This project aims at devoloping such catalyst materials at the nanoscale. In particular, Co@zeolite core-shell materials are synthesized and the effect of their structural architecture on the product distribution is evaluated.
Contact: M.Sc. Kerstin Wein
Collaborators: Prof. Dr. Jan-Dierk Grunwaldt, Prof. Dr. Krijn de Jong
Funding: Deutsche Forschungsgemeinschaft (DFG)
Neuronal Kinetics
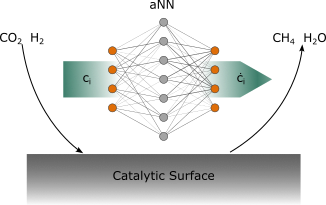
Methanation of carbon dioxide is a highly promising technology for both reducing local emissions of greenhouse gas and converting volatile renewable energy to easily storable and distributable methane gas. Thereby it provides the opportunity to couple gas and energy grid. However, various processes dominate the reactor kinetics on different length- and time-scales. Rigorous models describing all physical aspects of those interactions are scarce or even unfeasible. Nowadays the advent of cheap computational power and sophisticated methods in the fields of Machine Learning (ML) and artificial Neural Networks (aNNs) enables the description of complex phenomena through physically informed aNNs in multiple fields of research. Ultimately this project is aimed at describing the processes at the smallest length scales in methanation catalysts by the means of aNNs and coupling these with state-of-the-art reactor models for a flexible yet physically motivated grey-box approach to multiscale (micro-, meso- and macro-scale) reactor kinetics.
Contact: M.Sc. Hannes Stagge
Funding: German Federal Environmental Foundation (DBU)
Continuous Oxidation of Organics in a Gas-Liquid-Reactor
Gas-liquid reactions, such as hydrogenations or oxygenations of organic compounds, are a very important class of reactions in the chemical value chain. One of the major challenges is the mass transfer between gas and liquid phase in order to maximize the space-time-yield for the desired product. For continuous production in microstructured equipment segmented flow is preferable in many cases, as it achieves high mass transfer rates at moderate energy input. The project aims at continuous selective oxidation of an organic model compound with molecular polyoxometallate catalysts, with emphasis on derivation of the reaction kinetics and minimization of mass transfer issues ar the same time.
Contact: Dr.-Ing. Jens Friedland
Collaborators: Prof. Dr. Carsten Streb
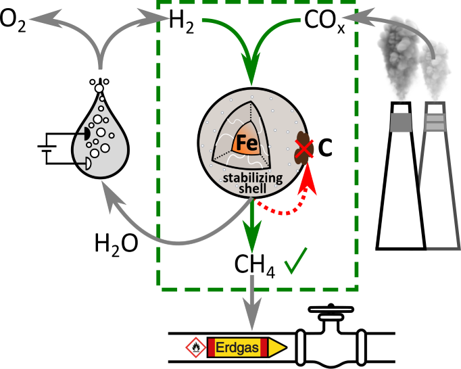
Iron-Based Core-Shell Model Catalysts for Methanation
Iron-based materials are environmently benign catalysts for syngas conversion into methane, which offer tolerance towards the feed gas mixture, due to their activity in both the CO/CO2 hydrogenation as well as the water-gas-shift reaction. Thus, catalysts based on abundantly available iron are highly interesting for utilizing industrial exhaust gases, consisting of mixtures of CO and CO2 in many cases associated with dynamic availability, for methanation. One of the major challenges for implementation of those materials are the complex transformations the iron-species undergo during reaction. Therefore, nanostructured Fe@SiO2 model catalysts uniquely offer to study those transformations and to develop a relation between material structure and catalytic perfomance, which is the basis for rational catalyst design.
Contact: M.Sc. Tobias Heinz
Collaborators: Prof. Dr. Sven Kureti
Ruthenium Catalysts for Fischer-Tropsch Synthesis
The project deals with the development of novel catalysts for Fischer-Tropsch synthesis for the decentralized liquefaction of atmospheric CO2 to 'green crude oil', which can subsequently be used as chemical feedstock. The goal is to reduce the reaction temperature window to below 150°C, in order to minimize the formation of undesirable gaseous by-products and thereby to simplify recycling of unconsumed feedstock. The optimized process will enable the development of simple reactor equipment suitable for mass production for a wide range of local applications in remote production sites.
Contact: M.Sc. Göran Baade
Funding: Vector Stiftung
Deactivation model development with machine learning
In industry, deactivation of solid catalysts contributes strongly to the lifetime costs of chemical plants. Predicting and reducing regeneration cycles can improve the productivity of a plant significantly which emphasizes the importance of deactivation modelling. Physical deactivation modelling requires a fundamental understanding of the processes at the catalyst scale. However, inference of deactivation mechanisms is difficult, why state-of-the-art models are mostly empirical. Our research aims at the development of deactivation models with high prediction capabilities without the need of comprehensive mechanistic understanding by the use of suitable machine learning methods.
Contact: M.Sc. Simon Hermann