I work as a research scientist at the Institute of Molecular Biology and Biotechnology of Prokaryotes at Ulm University. My team and I have extensive expertise in the physiology and metabolism of strictly anaerobic bacteria. At the moment, my research focuses on three main areas.
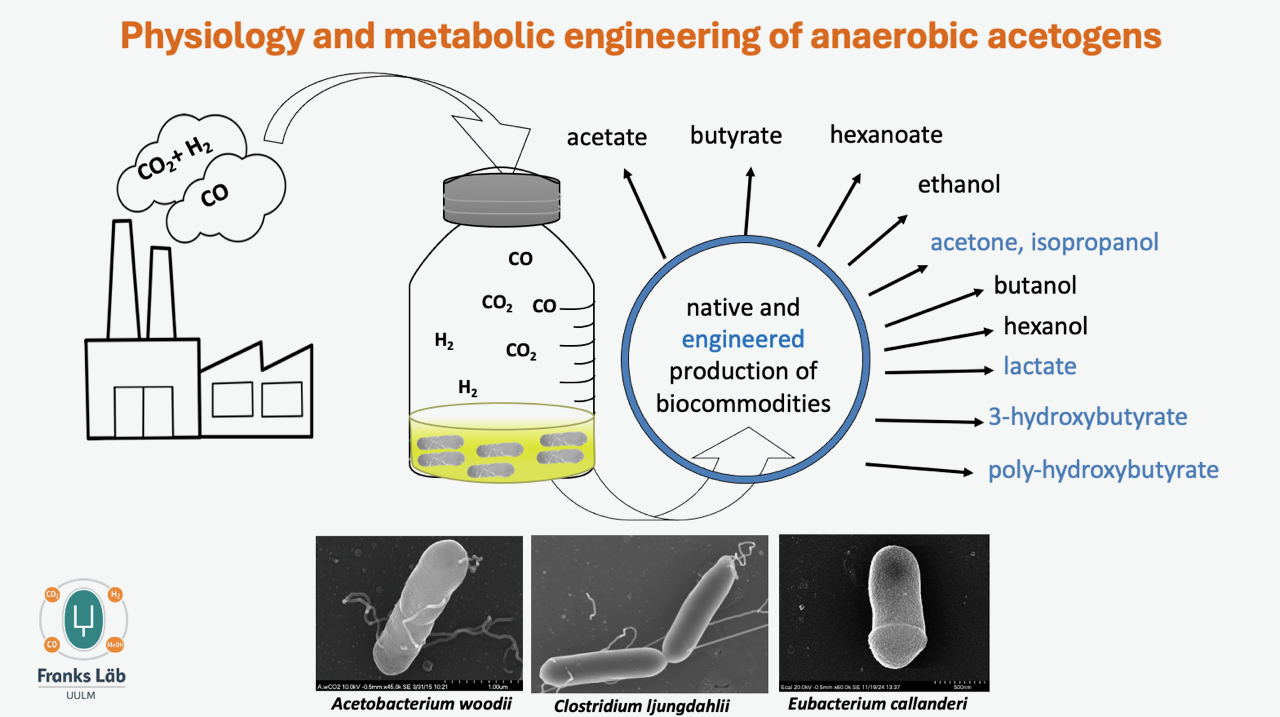
Metabolic engineering of the acetogenic bacteria Acetobacterium woodii, Clostridium ljungdahlii, and Eubacterium callanderii
Reducing CO₂ emissions is one of the most urgent global challenges in mitigating climate change. Anaerobic microbial conversion of CO₂ offers a sustainable way to produce chemicals without relying on fossil resources. Acetogenic bacteria use the Wood–Ljungdahl pathway for their anaerobic metabolism and can convert gaseous substrates such as CO or CO₂ + H₂ into products like acetate, butyrate, butanol, ethanol, caproate, hexanol, lactate, or 2,3‑butanediol—either through their natural metabolism or through engineered pathways.
Acetobacterium woodii, Clostridium ljungdahlii, and Eubacterium callanderii are important model organisms in our work. We often perform comparative physiological and genomic analyses of different acetogens to generate new hypotheses about their metabolism. These hypotheses are then tested by constructing recombinant strains, allowing us to gain new insights into their metabolic capabilities.
Anaerobic synthetic co‑cultures and sequential C1-fermentation
Microorganisms in nature live in complex communities, often forming metabolic relationships that can be mutualistic, commensal, or parasitic. In two DFG-funded projects (CaproMix and CarpoSyn), we studied the interactions between two specific anaerobic bacteria. Our goal was to develop a knowledge‑based, optimized synthetic co‑culture consisting of a genetically engineered A. woodii strain and a wild‑type strain of Clostridium drakei. In this system, A. woodii produces lactate and acetate from CO₂ + H₂, and C. drakei converts the lactate into butyrate (C4) and caproate (C6). We now apply this concept to other acetogenic strains—some with modified production traits—in combination with additional microorganisms to broaden the product spectrum.
Sequential C1-based fermentation processes consist of two physically separate bioprocess steps. In the first step, an acetogenic bacterium converts H₂ + CO₂, CO, methanol, or formate into acetate or lactate. In the second step, another microorganism uses these intermediates to produce higher‑value products. Depending on the process, either the entire cell suspension or only the spent medium from the first anaerobic fermentation is used for the second (usually aerobic) fermentation. This approach has been successfully demonstrated with A. woodii and Corynebacterium glutamicum to produce L‑glutamate, 3‑hydroxypropionate, or mevalonate from CO₂ via acetate. Other combinations include Ustilago maydis for triglyceride production and Pseudomonas putida for the synthesis of rhamnolipids.
Bacteria from anaerobic environments
Microbial communities in anaerobic habitats typically convert a wide range of substrates into acetate, CO₂, and H₂ or into methane. I am particularly interested in identifying and characterizing sofare unknown bacteria, especially those capable of using H₂ + CO₂ or producing compounds such as butyrate, caproate, or the corresponding alcohols.
For example, we successfully isolated and described a previously unknown anaerobic caproate‑producing bacterium. This organism was named Caproicibacter fermentans and is currently the only officially described species within this genus.
More about our ongoing projects see links below:
> MiMiWin II – Microorganisms as Helpers in Climate Protection (Funded by MWK-BW from 2025 to 2028)
> Microbiological Carbon Capture and Utilization – (MCCU ) as CO2-sink for wood coatings (BMWE funded from 2026 to 2029)
Our subproject: Development of genetically robust A. woodii strains for scaled lactate production from CO2 and H2
> CaproMix – Development of new and defined co-cultures for bioproduction of caproic acid from carbon dioxide (DFG funded from 2020 till 2023)
> CaproSyn - CO2-based production of caproic acid by engineered Acetobacterium woodii and Clostridium drakei, applying a synthetic co-culture (funded from 2023-2026)