A07: Role of myeloid-derived suppressor cells (MDSCs) in post-traumatic and post-septic immune responses
PI: G. Strauß
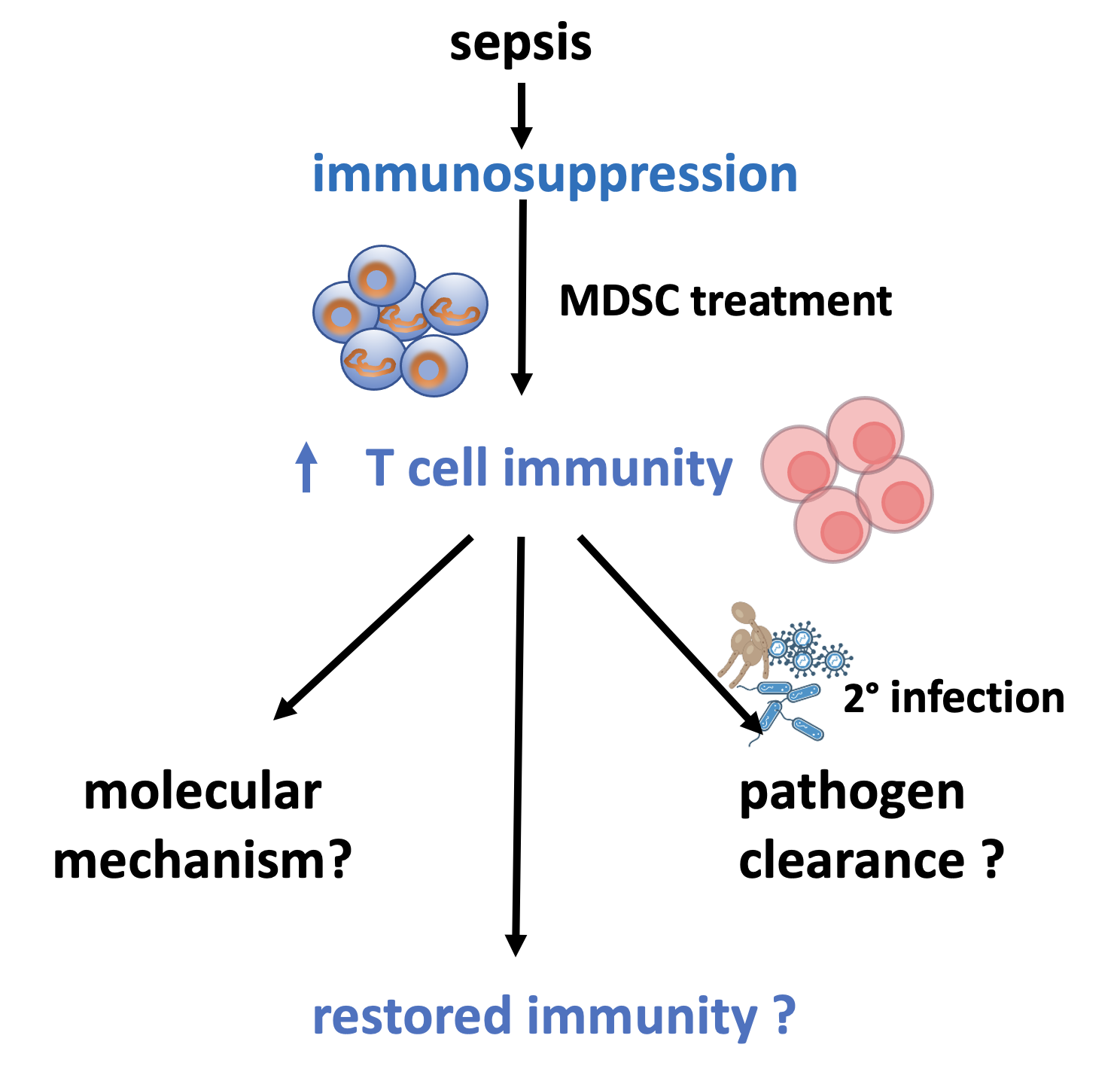
Severe trauma leads to long-lasting dysregulated adaptive immune responses with impaired T cell functions mediating immunosuppression and increased susceptibility for post-traumatic infections. Major regulators of T cells are myeloid-derived suppressor cells (MDSCs), which are initially described as immunosuppressive cells, but currently reported to have T cell stimulatory functions dependent on the pathological conditions. In the second funding period, we showed that a therapeutic adoptive transfer of in vitro-generated MDSCs into mice after blunt chest trauma (TxT) or sepsis induced by cecal ligation and puncture (CLP) activated and supported T cell functions by preventing post-traumatic T cell anergy and improving antigen-specific adaptive immune responses. As a direct follow up of our current project we wish to clarify in the third funding period 1. the molecular mechanisms underlying MDSC-based immunoactivation, 2. whether MDSC treatment protects the injured host against clinically relevant and frequently occurring post-traumatic viral, bacterial and fungal infections and 3. whether human in vitro-generated MDSCs can also improve post traumatic / septic T cell responses in humans. Our ultimate goal is to define whether in vitro-generated MDSCs have the potential to be used as a post-traumatic treatment option to improve T cell responses and counterbalance trauma-induced immunosuppression.
Projektleiterin
Prof. Dr. Gudrun Strauß
Universitätsklinik für Kinder- und Jugendmedizin
Forschungslabor
Eythstr. 24, Haus 16
89075 Ulm
Tel.: +49 731 500-57033
Fax: +49 731 500-57042
gudrun.strauss(at)uniklinik-ulm.de
Homepage