C02: Osteoblast differentiation-stage specific mechanisms of glucocorticoid action during fracture healing
PIs: J. Tuckermann, A. Ignatius
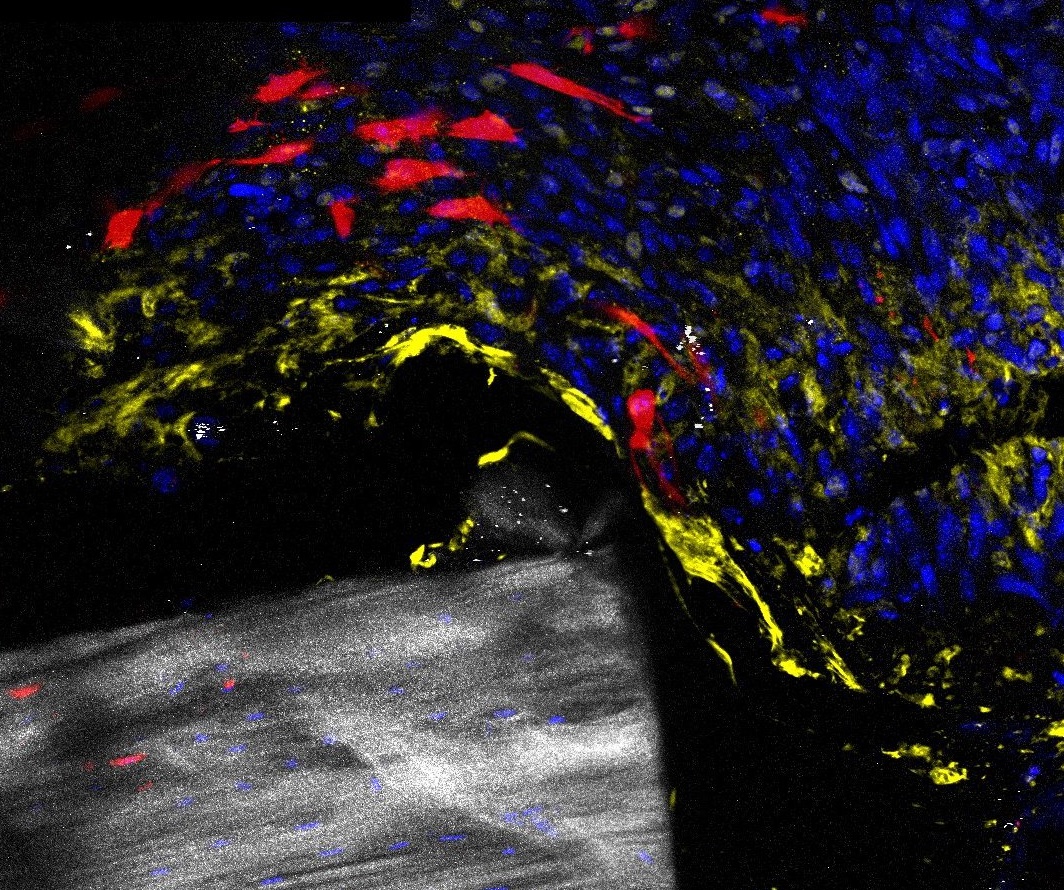
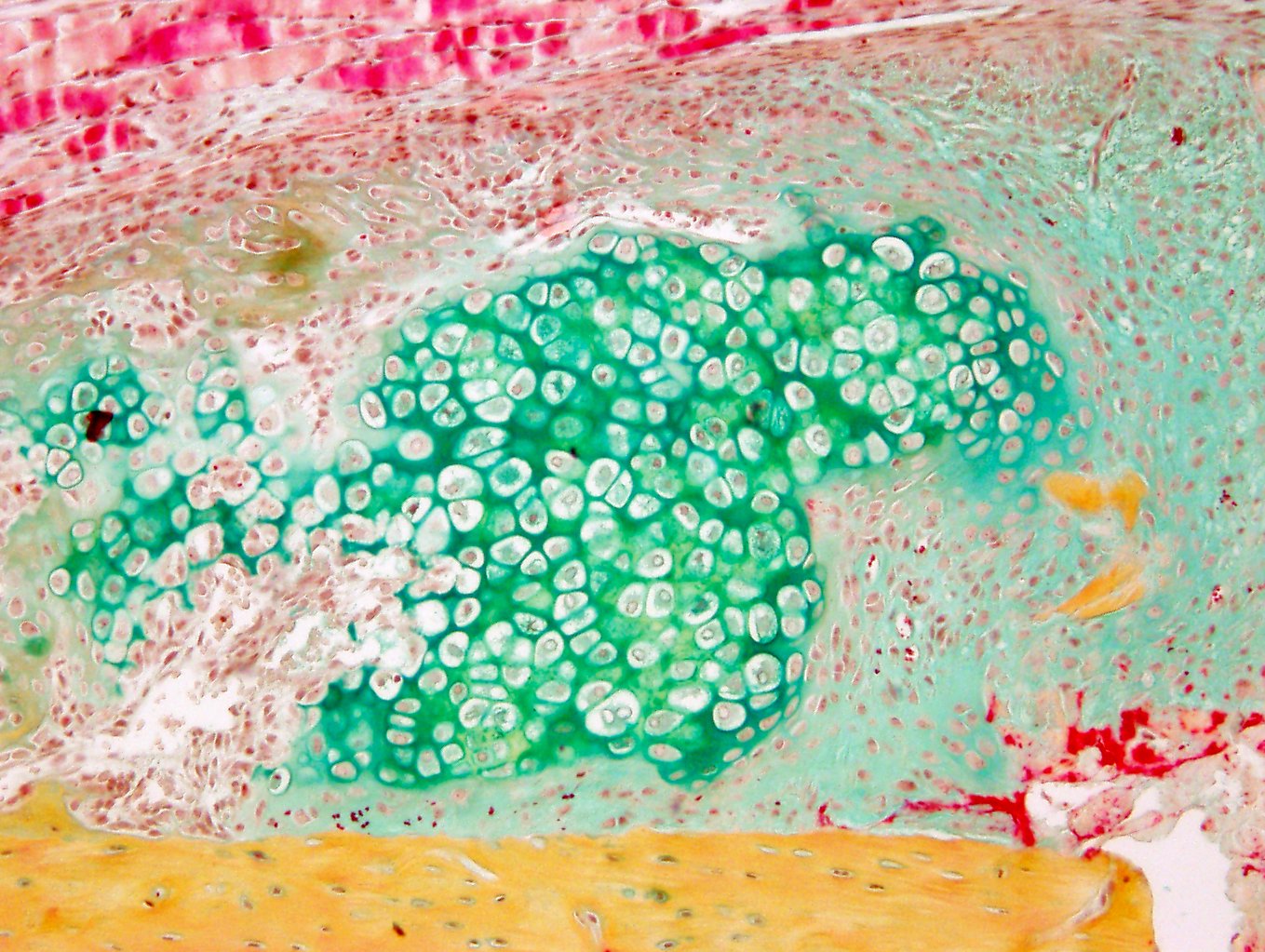
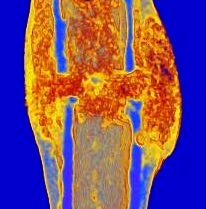
Glucocorticoids (GCs) are a major pillar of the stress response upon bone fracture, but also modulate the immune response and shape bone tissue homeostasis. We found that GCs via their receptor GR, but also GC counteracting factors, influence bone integrity and fracture healing. We have strong evidence that GR deletion in skeletal stem cells/progenitor cells (SSPCs), positive for Sox9+, increases bone mass and regeneration, whereas GR deletion in Osterix (Osx, Sp7)-labeled committed osteogenic cells results in opposite effects.Beyond GR’s role as a transcriptional activator, we discovered that it regulates cellular energy metabolism and, vice versa, energy metabolites influence GR activity. Therefore, C02 hypothesizes that 1) GR influences SSPCs differentially at distinct differentiation stages and 2) GR regulation of cellular energy metabolism affects differentiation and function of SSPCs and committed osteoblasts. Using lineage tracing, transcriptional profiling and via assessment of energy metabolism in skeletal stem cells/progenitor cells and committed osteoblasts, we will elucidate the stage-specific action of GR on cellular differentiation and cell metabolism during bone regeneration after fracture in close cooperation with projects A09N, B03, B09, C03, C08 and Z02. Our results will help to better understand the role of GC signaling in fracture healing, and thus form the basis for the development of new therapies for trauma patients with healing complications.
Projektleiter
Prof. Dr. Jan Tuckermann
Universität Ulm
Institut für Allgemeine Zoologie und Endokrinologie
Helmholtzstr. 8/1
89081 Ulm
Tel.: +49 731 500 32600
Fax: +49 731 500 32609
jan.tuckermann(at)uni-ulm.de
Prof. Dr. Anita Ignatius
Institut für Unfallchirurgische Forschung und Biomechanik
Zentrum für Traumforschung Ulm
Universitätsklinikum Ulm
Helmholtzstr. 14
89081 Ulm
Tel.: +49 731 500 55301
Fax: +49 731 500 55302
anita.ignatius(at)uni-ulm.de
www.biomechanics.de